Influenza, one of the most common infectious diseases, is a highly contagious airborne disease that appears as an acute febrile sickness with varying degrees of systemic symptoms ranging from mild fatigue to respiratory failure and death. Seasonal Influenza causes enormous workday loss, human suffering, and fatality with an annual death toll of ~300,000-650,000. Influenza virus infects multiple species and is a major pandemic threat.
Current Vaccines

Influenza vaccination is the preferred and most cost-effective intervention tool currently available to prevent influenza virus infection and disease. Licensed influenza vaccines include inactivated whole or split viruses, live attenuated vaccines, and recombinant viral subunit vaccines. Vaccines reduce influenza incidence, associated disease severity and mortality, yet their effectiveness remains moderate, especially for the elderly and individuals with co-morbidities. Most licensed vaccines are produced using embryonated hen’s eggs, which can lead to egg-induced adaptations that can potentially lower vaccine efficacy. In the Northern hemisphere, vaccines are typically supplied from September through the flu season which lasts until April.
Lead Candidates
Mynvax has developed improved seasonal vaccine candidates for protection against influenza. Current data from animal immunizations suggest that our recombinant subunit vaccines will be more efficacious compared to egg grown vaccines; additionally, they also confer protection to a wide range of influenza strains.
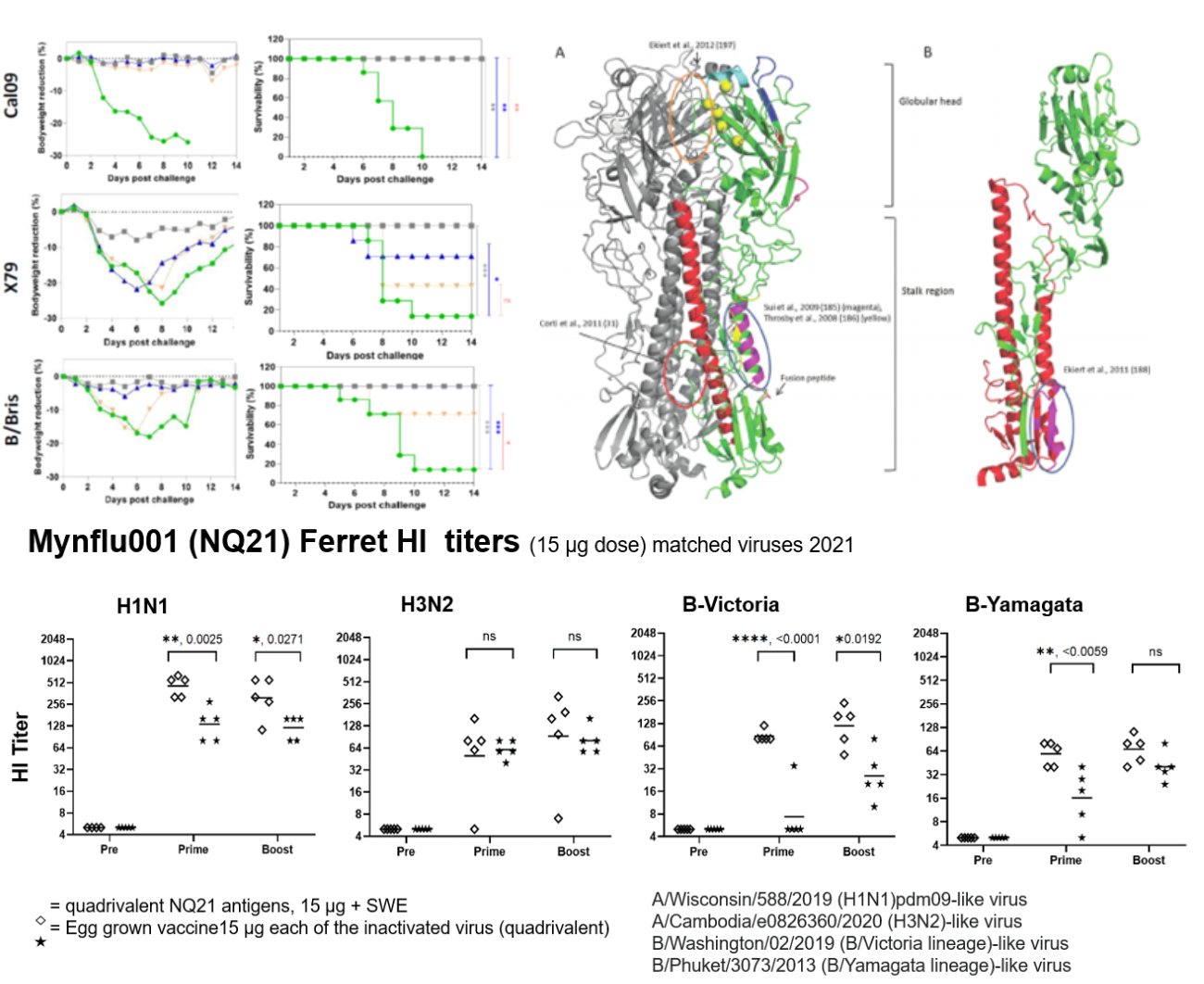
Mynflu001, is an adjuvanted quadrivalent recombinant subunit vaccine formulation containing Hemagglutinin derivatives of the seasonal WHO recommended strains with two formulations for the Northern and Southern Hemispheres respectively. Mynflu001 is manufactured in insect cells with good yields; a safety toxicity study in support of impending clinical trials in Australia is completed. Mynflu001 is entering clinical development in Australia and India.
Mynflu002, our pipeline candidate, is also an adjuvanted recombinant multivalent formulation which includes both the seasonal influenza vaccine antigens and additional novel, conserved antigens to enhance protection against antigenically drifted and shifted H1 and H3 strains. Clinical development of Mynflu002 will be implemented in the ENDFLU consortium supported by DBT, Government of India and Horizon 2020 funding from the European Union.
